why nmr?
|
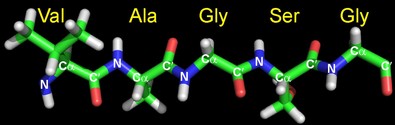
Atomic resolution
NMR spectra typically have very high spectral resolution. This means that peaks from virtually every single NMR-active nucleus (1H, 15N and 13C in isotope-labeled protein samples) can be individually resolved in protein NMR spectra. Since the list of NMR-active nuclei in proteins includes all the usual atoms except oxygen and sulphur, information on the structure and dynamics of the entire protein molecule can be obtained at atomic resolution.
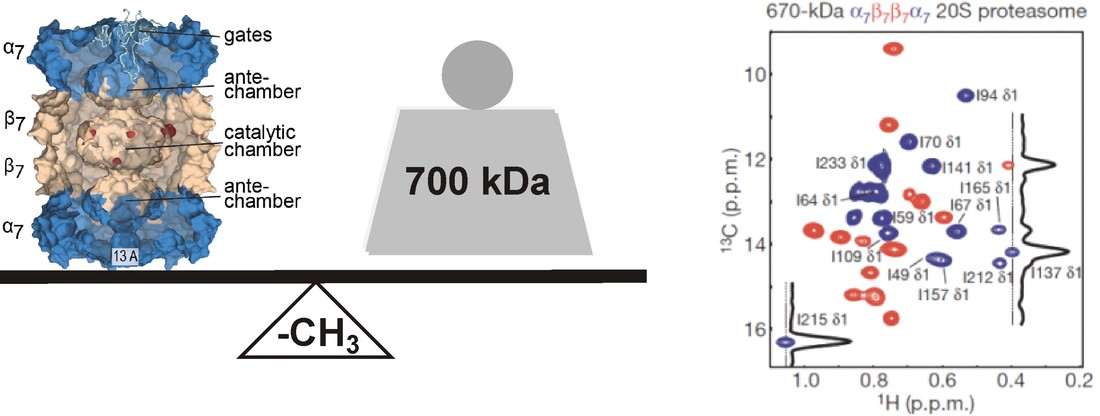
Methyls for large molecules upto 1 MDa
The favorable NMR properties of methyl groups can now be exploited to yield structural and dynamic information on large proteins and macromolecular complexes up to 1 MDa in size, such as the 20S proteasome in the above example.
|
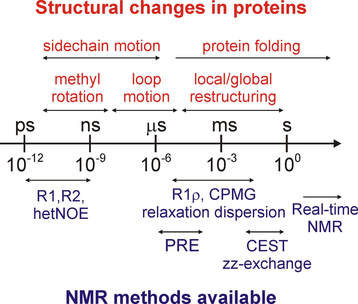
Comprehensive probe of protein dynamics
NMR methods are now available to obtain atomic-resolution information on protein motion occurring over a large range of timescales (picoseconds - seconds). This information enables us to build a complete and consistent picture of functional protein dynamics.
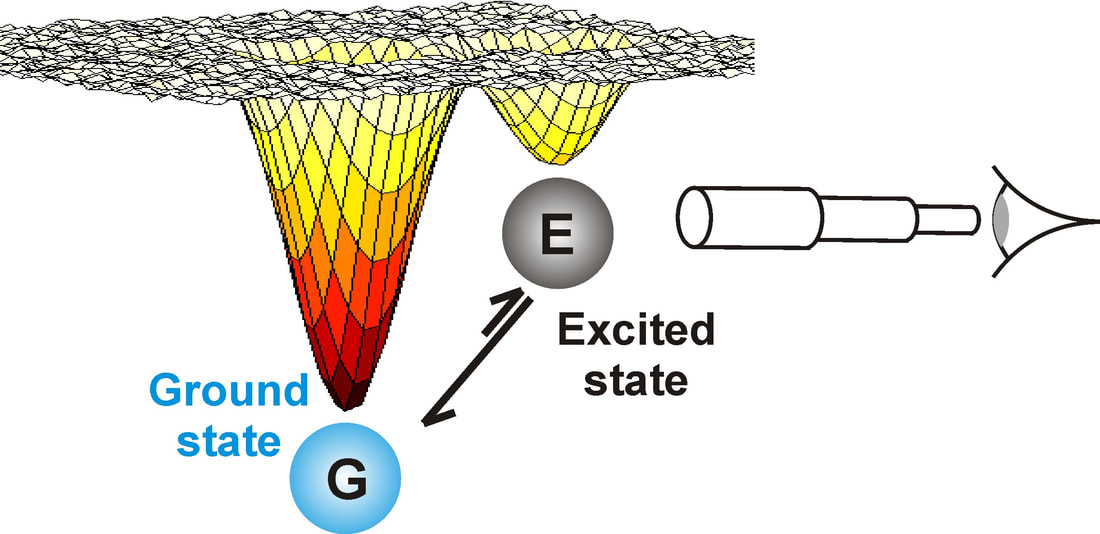
Studying sparsely populated structures
Recent methodological breakthroughs in NMR spectroscopy have also enabled the detection and structural characterization of higher energy protein conformations that are sparsely populated (~ 0.5 %) and invisible to traditional biophysical methods.
|